Technical Area
Please
click on one of the following links for a detailed introduction to
your chosen application :
Dissolved
Oxygen
Turbidity
pH
Conductivity
(EC)
Total Dissolved Solids (TDS)
ORP
(REDOX)
Salinity
Resistivity
Introduction
to Dissolved Oxygen
Introduction
Many gases mix with water like
nitrogen and oxygen without chemically reacting with it. Some gases
chemically react with water e.g. ammonia, CO2, and HCl. Oxygen does
not react with water. Dissolved Oxygen (DO) is really a physical
distribution of oxygen molecules in water. There are two main
sources of DO in water: atmosphere and photosynthesis. Waves and
tumbling water mix air into water where oxygen readily dissolves
until saturation occurs. Oxygen is also produced by aquatic plants
and algae as a by-product of photosynthesis.
Basic
Principle in DO measurement (OR What is Being Measured?)

The electrochemical method of
measuring DO requires a cathode, anode, electrolyte solution and a
gas permeable membrane. The material of the membrane is specially
selected to permit oxygen to pass through. Oxygen is consumed by the
cathode which will create a partial pressure across the membrane.
Oxygen will then diffuses into the electrolyte solution.
Thus, a DO meter actually measures
the pressure of oxygen in water. It can be used to measure DO in any
medium.
Polarographic
or Clark Cell Method
Dr. Clark first discovered the cell
to measure oxygen in 1956. This is basically an amperometric cell
that is polarized around 800 mV. This cell, named after Dr. Clark,
is built around the popular Ag/AgCl half-cell and a noble metal such
as gold, platinum or palladium. Reduction of oxygen is achieved
between 400 to 1200 mV, hence a need for a voltage of around 800 mV.
This is provided externally by a battery source.
Electrolyte used: KCl or KBr
| Cell |
Reaction |
| Anode |
2Ag
+ 2Cl- à 2AgCl + 2e- |
| Cathode
(Platinum, gold or palladium) |
2e-
+ ½ O2 + H2O à 2 OH- |
| Total
Reaction |
2e-
+ ½ O2 + H2O + 2Ag + 2Cl-
à 2 OH- + 2AgCl + 2e- |
From the above reaction, every time
oxygen is reduced at the cathode, 4 electrons or current is
generated directly proportional to the oxygen consumed (reduced) at
the cathode.
There are four major problems
associated with this type of DO measurement: .
| Problem |
Description |
| Isolation
of Anode |
Since
the net result of the chemical reaction is AgCl, over
time, a build up of AgCl will coat the anode. Once the
whole anode is covered, reaction stops and the oxygen
probe stops working. The probe can be reactivated by
cleaning the anode to remove the AgCl deposit which can be
a time-consuming procedure. |
| Zero
shift |
The
result of the above reaction produced more OH- ions which
will move the pH value of the electrolyte. The
electrolyte, which is around neutral pH value, will moves
into the alkaline range. This causes a zero shift, and
over time, the electrolyte will need to be changed. |
| Depletion
of Chloride |
The
net reaction also consumes Cl- ions. Over time, the
chloride ions will be consumed and the electrolyte needs
to be replaced. |
| Warm-up
Time |
The
major disadvantage is the need for an external power
source of approximately 800 mV to be applied to the
electrode. As soon as the probe is disconnected, power
supply is cut off. On connecting the probe again, the user
must wait for the probe to be polarized, that is, for the
current loop to be stabilized. This warm-up time is
approximately 10 minutes. Any measurement taken before
this warm-up time period will be normally a higher value
and will result in wrong readings. |
Galvanic
Cell Method
The galvanic probe principle was
introduced by Macreth in 1964 and has gone through a few changes.
The main advantage of a galvanic
probe is that is does not need an external power supply to provide
polarization as required by the Clark Cell. This is achieved by
using two dissimilar metals. In the presence of a electrolyte, there
is an electromotive voltage produced between the two metals. At
approximately 800 mV, this is large enough to reduce the oxygen at
the cathode. If lead and gold or lead and silver is used, the
differential voltage is approximately 800 mV.
Hence, a galvanic probe is really a
self-polarizing amperometric cell. The single biggest advantage is
the fact that the cell is now always ready and there is no warm up
time.
Electrolyte used: KCl or KBr
| Cell |
Reaction |
| Anode
(Zinc or Lead) |
Zn
à Zn2+ + 2e- |
| Cathode |
2e-
+ ½ O2 + H2O à 2OH- |
| Total
Reaction |
Zn
+ 2e- + ½ O2 + H2O à Zn2+
+ 2e- + 2OH-
Zn + ½ O2 + H2O
à Zn (OH)2
à ZnO (white
precipitate) + H2O or:
Zn + ½ O2 à
ZnO
|
Hence one molecule of oxygen
produces 4 electrons and there is a direct relationship between the
oxygen consumed at the cathode and the current produced by the cell.
The net result of the chemical
reaction is simply ZnO which is reasonably stable and does not coat
the anode. Water is recreated and the electrolyte is not consumed.
Theoretically, the electrolyte will go on forever without
replenishment.
Hence, because of the advantages
galvanic probe has, Eutech Instruments has chosen this superior
technology to make it easier for users.
DO
Measurement Considerations
The amount of DO that can be held
by water depends on 3 factors: water temperature, salinity, and
atmospheric pressure; Amount of DO increases with decreasing
temperature (colder water holds more oxygen).;Amount of DO increases
with decreasing salinity (freshwater holds more oxygen than
saltwater does);Amount of DO decreases with decreasing atmospheric
pressure (amount of DO absorbed in water decreases as altitude
increases).
The chart below shows the
solubility of DO in mg/l in water at various temperature.

Back to Tech Tips
Home
Introduction
to Turbidity Measurement
Definition
Turbidity is a cloudiness or haziness in water
(or other liquid) caused by individual particles that are too small
to be seen without magnification, thus being much like smoke in air.
Liquids can contain suspended solid matter consisting of particles
of many different sizes. While some suspended material will be large
enough and heavy enough to settle rapidly to the bottom of a
container if a liquid sample is left to stand (the settleable
solids), very small particles will settle only very slowly or not at
all if the sample is regularly agitated or the particles are
colloidal. These small solid particles cause the liquid to appear turbid.
There are several practical ways of quantifying cloudiness in
water, the most direct being some measure of attenuation (that is,
reduction in strength) of light as it passes through a sample column
of water. The now little-used Jackson Candle method (units: Jackson
Turbidity Unit or JTU) is essentially the inverse
measure of the length of a column of water needed to completely
obscure a candle flame viewed through it. The more water needed (the
longer the water column), the clearer the water. Of course water
alone produces some attenuation, and any substances dissolved in the
water that produce colour can attenuate some wavelengths. Modern
instruments do not use candles, but this approach of attenuation of
a light beam through a column of water should be calibrated and
reported in JTUs.
A property of the particles — that they will scatter a light
beam focused on them — is considered a more meaningful measure of
turbidity in water. Turbidity measured this way uses an instrument
called a nephelometer with the detector setup to the side of the
light beam. More light reaches the detector if there are lots of
small particles reflecting the source beam than if there are few.
The units of turbidity from a calibrated nephelometer are called
Nephelometric Turbidity Units (NTU). To some
extent, how much light reflects for a given amount of particulates
is dependent upon properties of the particles like their shape,
colour, and reflectivity. For this reason (and the reason that
heavier particles settle quickly and do not contribute to a
turbidity reading), a correlation between turbidity and TSS is
somewhat unique for each location or situation.
Turbidity in lakes, reservoirs, and the ocean can be measured
using a Secchi disk. This black and white disk is lowered into the
water until it can no longer be seen; the depth (secchi depth)is
then recorded as a measure of the transparency of the water
(inversely related to turbidity). The Secchi disk has the advantages
of integrating turbidity over depth (where variable turbidity layers
are present), being quick and easy to use, and inexpensive. It can
provide a rough indication of the depth of the euphotic zone with a
3-fold multiplication of the secchi depth. However, this
cannot be used in shallow waters where the disk can still be seen on
the bottom.
There are frequently standards on the allowable turbidity in
drinking water. In the United States (as of 2003) the allowable
standard is 1 NTU, with many drinking water utilities striving to
achieve levels as low as 0.1 NTU
Back to Tech Tips
Home
Introduction
to pH and pH Measurement
Definition
The pH of a solution measures the
degree of acidity or alkalinity relative to the ionization of water
sample. Pure water dissociates to yield 10-7 M of [H+]
and [OH-] at 25 oC; thus, the pH of water is
neutral i.e. 7.
pHwater
= - log [H+] = - log 10-7 = 7
Most pH readings range from 0 to
14. Solutions with a higher [H+] than water (pH less than
7) are acidic; solutions with a lower [H+] than water (pH
greater than 7) are basic or alkaline.

pH Measurement
Measuring pH involves comparing the
potential of solutions with unknown [H+] to a known
reference potential. pH meters convert the voltage ratio between a
reference half-cell and a sensing half-cell to pH values.
In acidic or alkaline solutions,
the voltage on the outer membrane surface changes proportionally to
changes in [H+]. The pH meter detects the change in
potential and determines [H+] of the unknown by the
Nernst equation:
E = Eo
+ (2.3RT)/nF log {unknown [H+]/internal [H+]}
where: E = total potential
difference (measured in mV); Eo = reference potential; R
= gas constant; T = temperature in Kelvin; n = number of electrons;
F = Faraday's constant; [H+] = hydrogen ion
concentration.
pH Temperature
Compensation
The pH of any solution is a
function of its temperature. Voltage output from the electrode
changes linearly in relationship to changes in pH, and the
temperature of the solution determines the slope of the graph. One
pH unit corresponds to 59.16 mV at 25 °C, the standard voltage and
temperature to which all calibrations are referenced. The electrode
voltage decreases to 54.20 mV/pH unit at 0.0 °C and increases to
74.04 mV/pH unit at 100.0 °C.
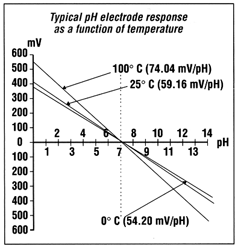
Since pH values are temperature
dependent, pH applications require some form of temperature
compensation to ensure standardized pH values. Meters and
controllers with automatic temperature compensation (ATC) receive a
continuous signal from a temperature element and automatically
correct the pH value based on the temperature of the solution.
Manual temperature compensation requires the user to enter the
temperature of the solution in order to correct pH readings for
temperature. ATC is considered to be more practical for most pH
applications.
pH System
A successful pH reading is
dependent upon all components of the system being operational.
Problems with any one of the three: electrode, meter or buffer will
yield poor readings.
pH Electrodes :
A pH electrode consists of two
half-cells; an indicating electrode and a reference electrode. Most
applications today use a combination electrode with both half cells
in one body. Over 90% of pH measurement problems are related to the
improper use, storage or selection of electrodes.
pH Meters :
A pH meter is a sophisticated volt
meter capable of reading small millivolt changes from the pH
electrode system. The meter is seldom the source of problems for pH
measurements. Today pH meters have temperature compensation (either
automatic or manual) to correct for variations in slope caused by
changes in temperature. Microprocessor technology has created many
new convenience features for pH measurement; auto-buffer
recognition, calculated slope and % efficiency, log tables for
concentration of ions and more.
pH Buffers :
These solutions of known pH value
allow the user to adjust the system to read accurate measurements.
For best accuracy:
- Standardization should be
performed with fresh buffer solutions.
- Buffer used should frame the
range of pH for the samples being tested.
- Buffers should be at the same
temperature as the samples. (For example: if all your samples
are at 50 °C, warm your buffers to 50 °C using a beaker in a
warm bath.)
Buffer values are dependent upon
temperature.
pH Electrode
Training Guide
Section 1: Electrode
Construction
The pH Sensitive Membrane
The most common type of sensitive membrane used on
a pH electrode is a blown glass bulb or rod. The glass used on Russell Mainstream
electrodes is suitable for most applications. If, however, the
application involves the constant monitoring of high temperature
liquids or high pH values (above pH 13), then an alternative glass
type can be specified. A bulb configuration will provide a fast
response and accurate results when used in a sample of low ionic
strength whereas a rod or bullet shaped membrane is very rugged and
will be more resistant to breakage.
The Reference Cell
Housed within the outer chamber of the pH
electrode is a reference system which is designed to provide a
stable reference voltage for the sensor. This reference 'half-cell'
will maintain a constant output in all liquids. Reference cells
consist of an internal element (usually a Ag/AgCl wire), an
electrolyte (usually KCl solution) and a liquid junction. The liquid
junction provides a leak path for the internal electrolyte to 'weep'
into the sample chamber and provide an electrical contact with the
liquid to be measured. If the liquid junction is not efficient then
measurements will be inaccurate.
The Cap/Cable/Connector
Electrodes used in laboratories are usually fitted
with 16mm diameter caps to suit cantilever arm electrode stands. The
cable used is a high grade, screened coaxial type with low noise
characteristics. Because of the high impedance of pH electrodes,
typically 100 megohms, connectors should always be kept clean and
dry. Detachable cable electrodes should not be used in very humid
environments.
Section 2: How to Specify an
Electrode
The following check list, when used with the
pH electrode selection chart, will help to identify a suitable
electrode for any given application.
- Sample Type
- Temperature
- Pressure
- Expected pH Range
- Viscosity of Sample
- Sample Volume
- Make and Model of pH Meter (to determine type
of connector)
- Cable Length Required
- Preferred Body Material (Glass or Plastic)
Section 3: Calibration of
pH Meter and Electrode
To achieve accurate, reproducible results a great
deal of attention needs to be paid to the calibration method. A
decision should be made on the accuracy required for the
measurement. This will enable the user to choose the type of
calibration required and the appropriate type of equipment to be
used. The following recommendations will ensure the best levels of
accuracy.
- All solutions should be stirred gently to
ensure the sensor is measuring a true representation of the
beaker contents.
- Calibration buffers should be chosen which have
pH values either side of the expected sample value, i.e, for a
sample which has an expected pH of 5, pH buffers with a value of
pH 7 and pH 4 should be used.
- Always use a 'control' buffer to keep a check
on the drift of the electrode. A method commonly used is to
place the electrode into a buffer, which has a value close to
the sample pH, between measurements.
- Fresh buffer solutions should be used. Changing
all solutions daily is a good practice.
- All solutions should be maintained at an equal
temperature.
- Rinse the electrode thoroughly in deionised
water between measurements.
- When calibrating the electrodes, allow
sufficient time to elapse for the reading to stabilise before
adjusting the meter. At least one minute, preferably longer.
Section 4: Procedure for
Calibrating the pH Meter
- 1 x high quality pH/mV meter
- 1 x 100ml pH 7.00 buffer solution
- 1 x 100ml pH 4.00 buffer solution
- 1 x 100ml pH 5.00 buffer solution
- 1 x calibrated glass thermometer
- 1 x temperature controlled water bath (required
if the sample value is different to ambient)
- 1 x combination pH electrode
- 4 x 200ml beakers
- 3 x Teflon stirrer paddles
- 1 x magnetic stirrer
- 1 x cantilever arm electrode stand
- 1 x fast flow wash bottle containing deionised
water
Method
- a) Assemble all equipment.
- b) Lower fill hole sleeve on the electrode (if
fitted) and thoroughly rinse the electrode tip.
- c) Lower electrode into gently stirred pH 7.00
buffer and allow to stabilise.
- d) Check the temperature of the calibration
solutions and adjust the default reading on the pH meter, if
applicable.
- e) After 1 - 2 minutes adjust the calibration
control on the pH meter to the appropriate pH value.
- f) Raise electrode from beaker and thoroughly
rinse with deionised water.
- g) Lower electrode into gently stirred pH 4.00
buffer and allow to stabilise.
- h) After 1 - 2 minutes adjust the slope control
on the pH meter to the appropriate temperature corrected value.
NOTE: Many modern microprocessor controlled pH
meters have automatic buffer recognition.
Please consult the instrument manual for specific adjustment
information.
- i) Rinse the electrode and repeat stages c) to
h) to confirm calibration.
- j) Rinse the electrode and lower into pH 5.00
buffer.
- k) After stabilising, record the reading in pH
5.00 buffer.
- l) Between measurements in the sample, rinse
and lower the electrode into the control buffer (pH 5.00) for
comparison with the recorded reading (remember to check
temperature pH versus pH values).
Section 5: Care and Maintenance
of Electrodes
By following this advice, it is possible to
significantly increase the expected life of an electrode and also to
improve the quality of measurement results.
- pH electrodes must always be stored wet. There
are many opinions on which storage solution is the best. Russell Mainstream
electrodes are all supplied soaked in a saturated KCl solution
with the exception of double junction electrodes which are
stored in the appropriate refill electrolyte for their
application.
- For short term storage, soak the electrode in
KCl.
- For long term storage, fill the soaking boot,
fit over the end of the electrode and seal with parafilm.
- Electrodes should never be stored in any of the
following liquids:
Deionised water, sample solutions, solvents, hydrochloric acid,
pH buffers containing mercury based preservatives.
- Sensing tips should always be rinsed after use.
- Reference cells should always be kept regularly
topped up with electrolyte.
- Connectors must be kept clean and dry.
- If the electrode needs to be cleaned
physically, always use soft tissue soaked in a mild detergent or
propanol.
- Regularly inspect the glass pH sensitive
membrane for cracks or chips.
Back to
Tech Tips
Home
Introduction
to Conductivity
Definition
Conductivity is the ability of a
material to conduct electric current. The principle by which
instruments measure conductivity is simple - two plates are placed
in the sample, a potential is applied across the plates (normally a
sine wave voltage), and the current is measured. Conductivity (G),
the inverse of resistivity (R) is determined from the voltage and
current values according to Ohm's law.
G = I/R = I
(amps) / E (volts)
Since the charge on ions in
solution facilities the conductance of electrical current, the
conductivity of a solution is proportional to its ion concentration.
In some situations, however, conductivity may not correlate directly
to concentration. The graphs below illustrate the relationship
between conductivity and ion concentration for two common solutions.
Notice that the graph is linear for sodium chloride solution, but
not for highly concentrated sulphuric acid. Ionic interactions can
alter the linear relationship between conductivity and concentration
in some highly concentrated solutions.

Units of
Measurement
The basic unit of conductivity is
the siemens (S), formerly called the mho. Since cell geometry
affects conductivity values,  standardized
measurements are expressed in specific conductivity units (S/cm) to
compensate for variations in electrode dimensions. Specific
conductivity (C) is simply the product of measured conductivity (G)
and the electrode cell constant (L/A), where L is the length of the
column of liquid between the electrode and A is the area of the
electrodes (see Figure 1). standardized
measurements are expressed in specific conductivity units (S/cm) to
compensate for variations in electrode dimensions. Specific
conductivity (C) is simply the product of measured conductivity (G)
and the electrode cell constant (L/A), where L is the length of the
column of liquid between the electrode and A is the area of the
electrodes (see Figure 1).
C = G x (L/A)
If the cell constant
is 1 cm-1, the specific conductivity is the same as the measured
conductivity of the solution. Although electrode shape varies, an
electrode can always be represented by an equivalent theoretical
cell.
The following table
shows optimum conductivity ranges for cells of three different
constants:
| Cell
constant |
Optimum
Conductivity Range (µS/cm) |
| 0.1 |
0.5
to 400 |
| 1.0 |
10
to 2000 |
| 10.0 |
1000
to 200,000 |
Conductivity
Temperature Compensation
Conductivity
measurements are temperature dependent. The degree to whcih
temperature affects conductivity varies from solution to solution
and can be calculated using the following formula:
Gt = Gtcal
{1 + a(T-Tcal)}
where: Gt =
conductivity at any temperature T in °C, Gtcal =
conductivity at calibration temperature Tcal in °C, a =
temperature coefficient of solution at Tcal in °C.
| Substance
at 25°C |
Concentration |
Alpha
(a) |
| HCl |
10
wt% |
1.56 |
| KCl |
10
wt% |
1.88 |
| H2SO4 |
50
wt% |
1.93 |
| NaCl |
10
wt% |
2.14 |
| HF |
1.5
wt% |
7.20 |
| HNO3 |
31
wt% |
31.0 |
Common alphas (a) are
listed in the table above. To determine that a of other solutions,
simply measure conductivity at a range of temperatures and graph the
change in conductivity versus the change in temperature. Divide the
slope of the graph by Gtcal to get a.
All meters have either
fixed or adjustable automatic temperature compensation referenced to
a standard temperature - usually 25°C. Most meters with fixed
temperature compensation use a a of 2%/°C (the approximate a of
NaCl solutions at 25°C). Meters with adjustable temperature
compensation let you to adjust the a to more closely match the a of
your measured solution.
Conductivity
Meter Calibration and Cell Maintenance
Conductivity meters
and cells should be calibrated to a standard solution before using.
When selecting a standard, choose one that has the approximate
conductivity of the solution to be measured. The conductivity of
some common solutions is shown in the table below.
| Solution |
Conductivity |
| Absolute
pure water |
0.055
µS/cm |
| Power
plant boiler water |
1.0
µS/cm |
| Good
city water |
50
µS/cm |
| Ocean
water |
53
mS/cm |
A polarized or fouled
electrode must be cleaned to renew the active surface of the cell.
In most situations, hot water with a mild liquid detergent is an
effective cleanser. Acetone easily cleans most organic matter, and
chlorous solutions will remove algae, bacteria or molds. To prevent
cell damage, abrasives or sharp objects should not be used to clean
an electrode. A cotton bud works well for cleaning but care must be
taken not to widen the distance of cell.
Back
to Tech Tips
Home
Introduction to Total
Dissolved Solids (TDS)
Total Dissolved Solids (TDS) are solids in water
that can pass through a filter. TDS is a measure of the amount of
material dissolved in water. This material can include carbonate,
bicarbonate, chloride, sulphate, phosphate, nitrate, calcium,
magnesium, sodium, organic ions, and other ions. A certain level of
these ions in water is necessary for aquatic life. Changes in TDS
concentrations can be harmful because the density of the water
determines the flow of water into and out of an organism's cells
(Mitchell and Stapp, 1992). However, if TDS concentrations are too
high or too low, the growth of many aquatic life can be limited, and
death may occur.
Similar to TSS, high concentrations of TDS may
also reduce water clarity, contribute to a decrease in
photosynthesis, combine with toxic compounds and heavy metals, and
lead to an increase in water temperature. TDS is used to estimate
the quality of drinking water, because it represents the amount of
ions in the water. Water with high TDS often has a bad taste and/or
high water hardness, and could result in a laxative effect.
The TDS concentration of a water sample can be
estimated from specific conductance if a linear correlation between
the two parameters is first established. Depending on the chemistry
of the water, TDS (in mg/l) can be estimated by multiplying specific
conductance (in micromhos/cm) by a factor between 0.55 and 0.75.
TDS can also be determined by measuring individual
ions and adding them up.
Back
to Tech Tips
Home
Introduction
to ORP (REDOX)
Oxidation-Reduction Potential (ORP)
or Redox potential measurements are used to monitor chemical
reactions, to quantify ion activity, or to determine the oxidizing
or reducing properties of a solution. ORP is a measurement of the
electrical potential of a redox reaction and serves as a yardstick
to judge how much oxidation or reduction takes place under existing
conditions.
ORP electrodes measure the voltage
across a circuit formed by the measuring metal half cell and the
reference half cell. When the ORP electrode is placed in the
presence of oxidizing or reducing agents, electrons are constantly
transferred back and forth on its measuring surface, generating a
tiny voltage. The ORP measurement can be made using the millivolt
mode of a pH meter.
ORP measurement may be utilized
very successfully in many commercial and industrial applications.
These include:
-
Cyanide
Oxidation
-
Aquarium
Monitoring
-
Chromate
Reduction
-
Drinking Water
-
Swimming Pool
Water
-
Pulp Bleaching
-
Cooling Tower
-
Ozone
Monitoring
-
Water
Pollution Monitoring
ORP technology has been gaining
recognition worldwide and is found to be a reliable indicator of
bacteriological water quality for sanitation - determine free
chlorine parameter. In swimming pool application, the ideal ORP
value is approximately 700 mV where the Kill Time of E.Coli bacteria
is the fastest to ensure good water quality. However ORP value also
depends on the pH of pool water, which is typically between 7.2 and
7.6 pH.

The pH of pool water has to be
maintained at optimum level by dosing appropriate chemicals. If the
pH of swimming pool is acceptable and ORP value is below 700 mV,
then hypochlorite or other oxidizing chemicals need to be added.
Back
to Tech Tips
Home
Introduction
to Salinity
Salinity refers to the concept of saltiness of a
body of water. Water is defined as saline if it contains 3 to 5%
salt by volume. The ocean is naturally saline at ~3.5% salt (see sea
water). Some inland salt lakes (seas) are even saltier. The
technical term for ocean saltiness is halinity, from the
fact that halides (chloride, specifically) are the most abundant
anion in the mix of dissolved solids. In oceanography, it has been
traditional to express salinity as concentration in parts per
thousand (ppt or o/oo), which is grams salt per litre of
water.
The salt content of most lakes, rivers, and streams is so small
that these waters are termed fresh or even sweet water.
Salt is difficult to remove from water, and salt content is a factor
in water potability. Salinity is an ecological factor of
considerable import, influencing the types of organisms that live in
a body of water. As well, salinity influences the kinds of plants
that will grow either in a water body, or on land fed by a water (or
by a groundwater). A plant adapted to a saline conditions is called
a halophyte (for salt loving). See also biosalinity.
Animals and bacteria that can live in very salty conditions are
classified as extremophiles.
Systems of Classification
of water bodies based upon Salinity
Marine waters are those of the ocean, another term for which is euhaline
seas. The salinity range for euhaline seas is 30 to 35 o/oo.
brackish seas or waters have salinity in the range
of 0.5 to 29 o/oo; and metahaline seas
from 36 to 40 o/oo. These waters are all grouped as homoiohaline
because their salinity is derived from the ocean (thalassic)
and essentially invariant, in contrast to poikilohaline environments
in which the salinity variation is biologically significant (Dahl,
1956).
Poikilohaline waters may range anywhere from 0.5
o/oo to greater than 300 o/oo. The important
characteristic is that these waters tend to vary in salinity over
some biologically meaningful range seasonally or on some other
roughly comparable time scale. Put simply, these are bodies of water
with variable salinity. The following table, modified from Por
(1972) follows the "Venice system" (1959):
| |
THALASSIC SERIES |
| >300 o/oo |
-------------------- |
| |
hyperhaline |
| 60 - 80 o/oo |
-------------------- |
| |
metahaline |
| 40 o/oo |
-------------------- |
| |
mixoeuhaline |
| 30 o/oo |
-------------------- |
| |
polyhaline |
| 18 o/oo |
-------------------- |
| |
mesohaline |
| 5 o/oo |
-------------------- |
| |
oligohaline |
| 0.5 o/oo |
-------------------- |
Highly saline water is referred to as brine.
- Dahl, E. 1956. Ecological salinity boundaries in poikilohaline
waters. Oikos, 7(I): 1–21.
- Por, F. D. 1972. Hydrobiological notes on the high-salinity
waters of the Sinai Peninsula. Mar. Biol., 14(2):
111–119.
- Venice system. 1959. Final resolution of the symposium on the
classification of brackish waters. Archo Oceanogr. Limnol.,
11 (suppl): 243–248.
Back
to Tech Tips
Home
Electrical
Resistivity Definition
Electrical resistivity
(also known as specific electrical resistance)
is a measure indicating how strongly a material opposes
the flow of electric current. A low resistivity
indicates a material that readily allows the movement of
electrons. The SI unit for electrical resistivity is the
ohm metre.
The electrical resistivity of a
material is usually given by :

where
ρ is
the electrical resistivity (measured in ohm metres)
R is
the resistance of a uniform specimen of the material
(measured in ohms)
l is
the length of the specimen (measured in metres)
S is
the cross-sectional area of the specimen (measured in
square metres)
Electrical resistivity can also be defined as:

where
E is
the magnitude of the electric field (measured in volt
metres)
J is
the magnitude of the current density (measured in
amperes per square metre)
In general, electrical resistivity of
metals increases with temperature, while the resistivity
of semiconductors decreases with temperature. As the
temperature of a metal is reduced, the resistance
usually reduces until it reaches a constant value, known
as the residual resistivity. This value depends not only
on the type of metal, but on its purity and thermal
history. Some materials lose all electrical resistivity
at sufficiently low temperatures, due to an effect known
as superconductivity.
The reciprocal quantity is electrical
conductivity.
Typical values
Typical resistivities for various
materials (at 20 °C; 10-6 Ωm equals Ω·mm²/m)
are shown in the table below:
| Material |
Resistivity (ohm metres) |
| Silver |
0.0159 × 10-6 |
| Copper |
0.017 × 10-6 |
| Gold |
0.0244 × 10-6 |
| Aluminium |
0.0282 × 10-6 |
| Tungsten |
0.056 × 10-6 |
| Iron |
0.1 × 10-6 |
| Steel, Stainless |
0.72 × 10-6 |
| Platinum |
0.11 × 10-6 |
| Lead |
0.22 × 10-6 |
Nichrome
(A nickel-chromium alloy commonly
used in heating elements) |
1.50 × 10-6 |
| Carbon |
35 × 10-6 |
| Germanium |
0.46 |
| Silicon |
640 |
| Glass |
1010 to 1014 |
| Hard rubber |
approximately 1013 |
| Sulfur |
1015 |
| Quartz (fused) |
75 × 1016 |
Back
to Tech Tips
|